Aging revolves around cellular senescence, a state of cell arrest. Extreme stress levels or cell proliferation can lead to the accumulation of senescent cells, which limits the capacity of tissues to function and regenerate in aged organisms. Nicotinamide phosphoribosyltransferase (Nampt) is a crucial enzyme incorporated in cellular senescence, and it plays a vital role in synthesizing a critical coenzyme called nicotinamide adenine dinucleotide (NAD+), which contributes to health, aging, and cellular metabolism. Current research shows that cells obtained from genetically altered mice with artificially stimulated Nampt were tolerant to senescence induced by excessive cell proliferation; however, we have to determine whether these cells are protected from stress-activated senescence.
In an article published in the journal Genes to Cells, a team of researchers led by investigators from the Nara Institute of Science and Technology in Japan demonstrated that cells from genetically altered mice with artificially stimulated Nampt were resilient to premature stress-activated senescence in a laboratory dish. “Our results demonstrate that NAMPT/NAD+ functions to protect cells not only from replicative senescence but also from stress-induced premature senescence,” said the investigators in their publication. The investigators suggested that stimulation of NAMPT activity or elevations in NAD+ levels would prevent tissues from acquiring premature non-replicating cells, thus facilitating healthy aging.
Cellular stress is the primary cause of aged organisms accumulating these senescent cells. Various forms of stress lead to premature senescence in cells. One form is oxidative stress, which occurs upon an improper ratio between deleterious compounds called free radicals and supportive antioxidants in the body. In addition, the cell structure called the endoplasmic reticulum is also responsible for inducing stress through the accumulation of irregular proteins. Furthermore, the accumulation of irregular proteins stimulates the unfolded protein response (UPR), a cellular repair mechanism that removes irregular proteins and improves the health and function of cells.
Studies have demonstrated that senescent human and mouse cells exhibit reduced NAMPT followed by NAD+. Artificial stimulation of NAMPT induces elevated cellular NAD+ levels, thereby halting onset senescence in cells. However, excessive cell proliferation is what promoted the effects seen in these senescent cells, not stress. Because of this, the investigators from the Nara Institute of Science and Technology evaluated the function of NAMPT in cells experiencing senescence stimulated by stress.
To conduct this experiment, the investigators utilized oxidative and ER stressors to stimulate premature senescence in mouse cells that were genetically altered to have an artificially inserted stimulated form of Nampt. When the cells were exposed to an oxidative stressor, the team of researchers noticed that cells with artificially stimulated Nampt exhibited more division and multiplication than unmodified cells. In addition, the cells with artificially stimulated Nampt were found to be less senescent than the unmodified cells after treatment with an oxidative stressor. Thus, the presented findings suggest that artificial stimulation of NAMPT prevents mouse cells from undergoing premature senescence caused by oxidative stress.
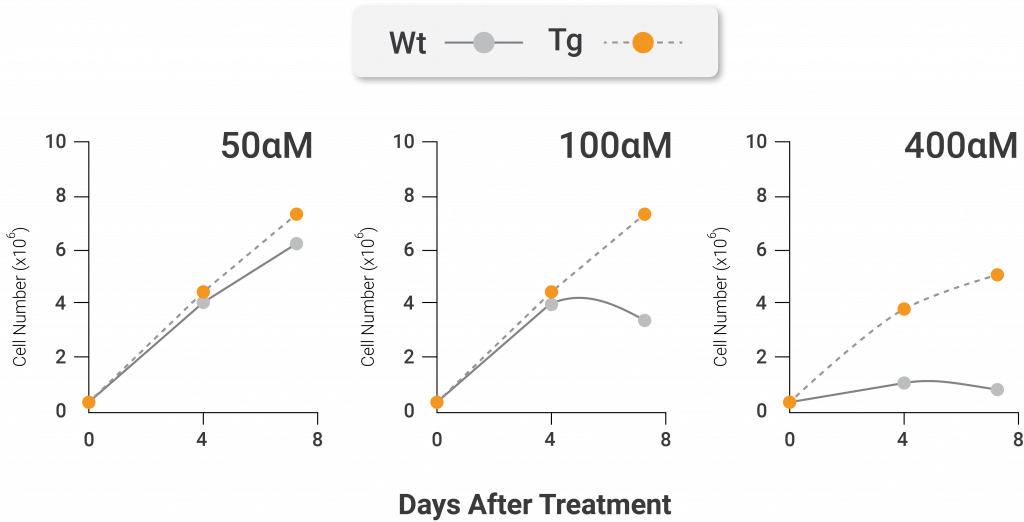
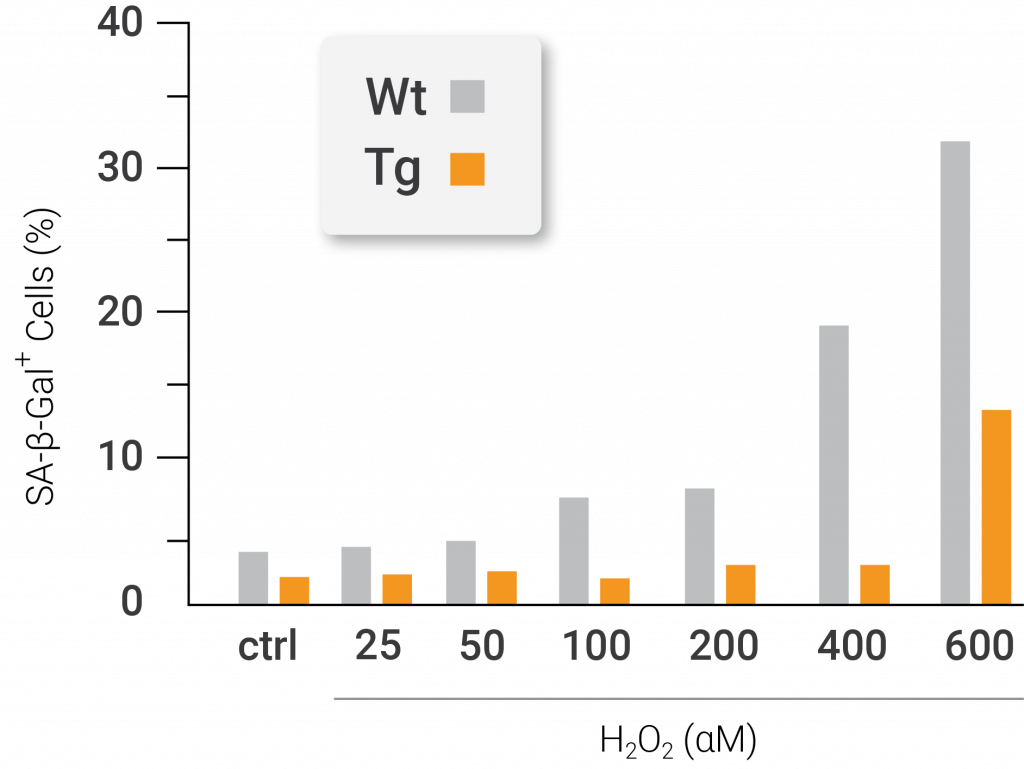
Nampt protects against oxidative stress-induced premature senescence. The total cell number (left) and percentage of senescent positive cells (right) after treatment with the oxidative stressor hydrogen peroxide (H2O2). The cells from genetically modified mice containing artificially active nicotinamide phosphoribosyltransferase (Nampt) show higher total cell numbers and less senescent cells following treatment with the oxidative stressor compared to cells that come from unaltered mice. The white lines and bars are indicative of unaltered mice. The black lines and bars are indicative of genetically modified mice with artificially activated Nampt (Nuriliani Genes to Cells | 2020).
After cells with artificially stimulated Nampt underwent treatment with an ER stressor, investigators noticed that the experimental results were similar to the oxidative stress treatment. These treated cells also underwent more replication and displayed less senescence than the unmodified cells that were treated with an ER stressor. Moreover, the cells with artificially stimulated Nampt better circumvented cellular senescence by promoting the UPR gene program more than unmodified cells.
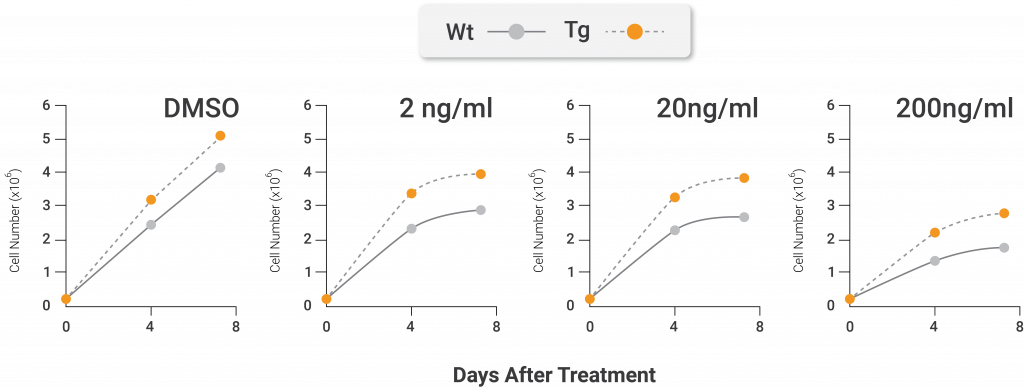
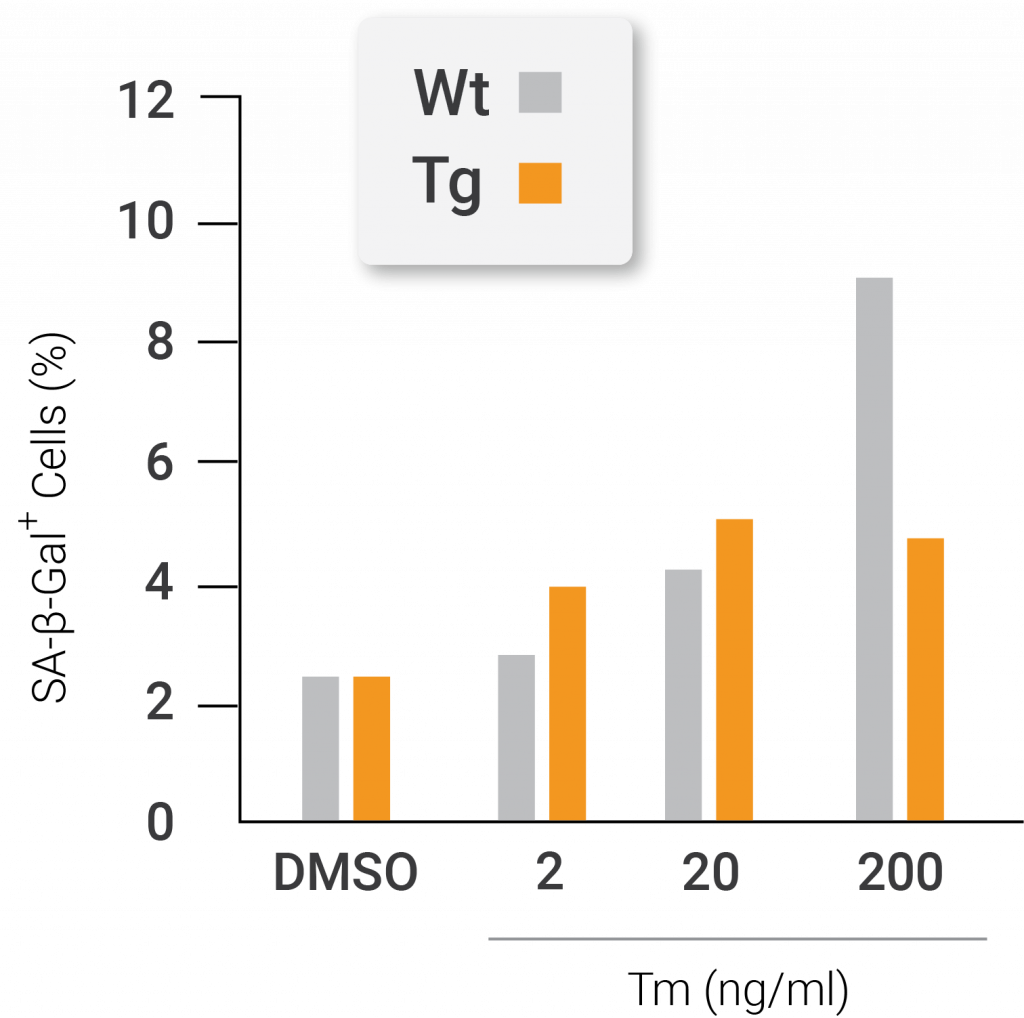
Nampt protects against endoplasmic reticulum stress-induced premature senescence. The total cell number (left) and percentage of senescent positive cells (right) after treatment with the endoplasmic reticulum (ER) stress-inducing agent tunicamycin. The cells from genetically modified mice containing artificially active nicotinamide phosphoribosyltransferase (Nampt) show higher total cell numbers and less senescent cells following treatment with the oxidative stressor compared to cells that come from unaltered mice. The white lines and bars are indicative of unaltered mice. The black lines and bars are indicative of genetically modified mice with artificially activated Nampt (Nuriliani Genes to Cells | 2020).
“Together with our previous and current reports, we were able to show that activation of the NAMPT/NAD+ axis acts as protection against both replicative senescence and stress-induced premature senescence,” said the investigators. Furthermore, the investigators believe that this study could expedite research concerning both the utilization of NAD+ precursors and the generation of NAMPT active molecules to shield tissues from accumulating premature senescent cells, thus preserving healthy aging.